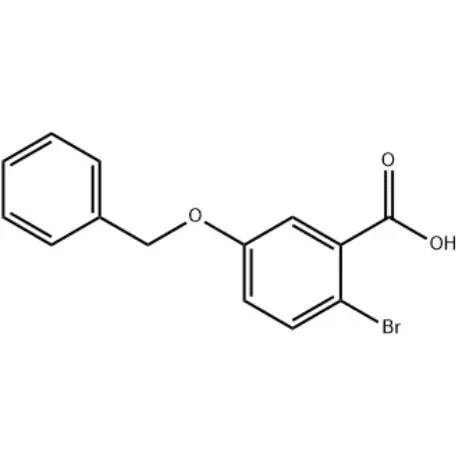
In the world of organic chemistry, the discovery and development of new compounds play a crucial role in advancing various fields, including pharmaceuticals, materials science, and agrochemicals. One such compound that has gained significant attention is 5-Benzyloxy-2-bromo-benzoic acid. With its unique structure and properties, this compound has found diverse applications in different industries. In this blog post, SACH will share the various uses and potential of 5-Benzyloxy-2-bromo-benzoic acid, shedding light on its importance and impact in the scientific community.
Applications of 5-Benzyloxy-2-Bromo-Benzoic Acid for sale
1. Synthesis of Biologically Active Compounds:
One of the primary applications of 5-Benzyloxy-2-bromo-benzoic acid lies in its role as a key intermediate in the synthesis of biologically active compounds. This compound serves as a building block for the preparation of various pharmaceutical agents, including anti-inflammatory drugs, antiviral agents, and anticancer compounds. The presence of the bromine and benzyl groups in its structure allows for further modifications, enabling the creation of novel molecules with enhanced therapeutic properties.
2. Agrochemical Development:
The agricultural industry heavily relies on the development of effective agrochemicals to protect crops from pests and diseases. 5-Benzyloxy-2-bromo-benzoic acid has emerged as a valuable tool in this area. By incorporating this compound into the synthesis of agrochemicals, researchers have been able to enhance the efficacy and selectivity of these products. Its unique structure and reactivity make it an ideal candidate for the development of herbicides, fungicides, and insecticides, contributing to sustainable agriculture practices.
3. Material Science and Polymer Chemistry:
The field of material science constantly seeks new compounds that can be used in the development of advanced materials with improved properties. 5-Benzyloxy-2-bromo-benzoic acid has proven to be a valuable asset in this regard. Through its incorporation into polymer chains, it imparts desirable characteristics such as thermal stability, mechanical strength, and chemical resistance. This compound has been utilized in the synthesis of polymers for applications in coatings, adhesives, and electronic devices, opening up new possibilities for material engineering.
4. Catalysts and Ligands:
Catalysts and ligands play a crucial role in various chemical reactions, enabling the transformation of reactants into desired products. 5-Benzyloxy-2-bromo-benzoic acid has shown promise as a catalyst or ligand in several organic transformations. Its unique structure allows for the coordination of transition metals, facilitating catalytic processes such as cross-coupling reactions, C-H activation, and asymmetric synthesis. The versatility of this compound in catalysis has attracted significant attention from researchers, leading to the development of new methodologies and strategies in organic synthesis.
5. Research and Development:
Apart from its direct applications, 5-Benzyloxy-2-bromo-benzoic acid has become a valuable tool in research and development. Its availability and ease of synthesis make it a popular choice for scientists exploring new chemical reactions and methodologies. By utilizing this compound as a starting material or intermediate, researchers can explore its reactivity and potential for creating novel compounds with unique properties. This compound has become an integral part of the toolkit for synthetic chemists, enabling the discovery of new molecules and the advancement of scientific knowledge.
Conclusion:
In conclusion, 5-Benzyloxy-2-bromo-benzoic acid has emerged as a versatile compound with diverse applications in various scientific fields. Its unique structure and reactivity make it an invaluable tool for the synthesis of biologically active compounds, the development of agrochemicals, the advancement of material science, and the exploration of catalytic processes. Furthermore, its availability and ease of synthesis have made it a popular choice for researchers in their quest for new chemical reactions and methodologies. As the scientific community continues to uncover the potential of this compound, we can expect further advancements and discoveries that will shape the future of chemistry and its applications.
https://www.hzsqchem.com/Applications-of-5-Benzyloxy-2-bromo-benzoic-acid.html
SACH
sales@hzsqchem.com

